Estimated read time: 3-4 minutes
This archived news story is available only for your personal, non-commercial use. Information in the story may be outdated or superseded by additional information. Reading or replaying the story in its archived form does not constitute a republication of the story.
SALT LAKE CITY — More than 615,800 Utahns know what it feels like to be tested for COVID-19.
They know it is somewhat invasive, involving a long swab inserted up the nose and reaching to the back of the throat where it stays for several potentially uncomfortable seconds.
But it doesn’t have to be that way.
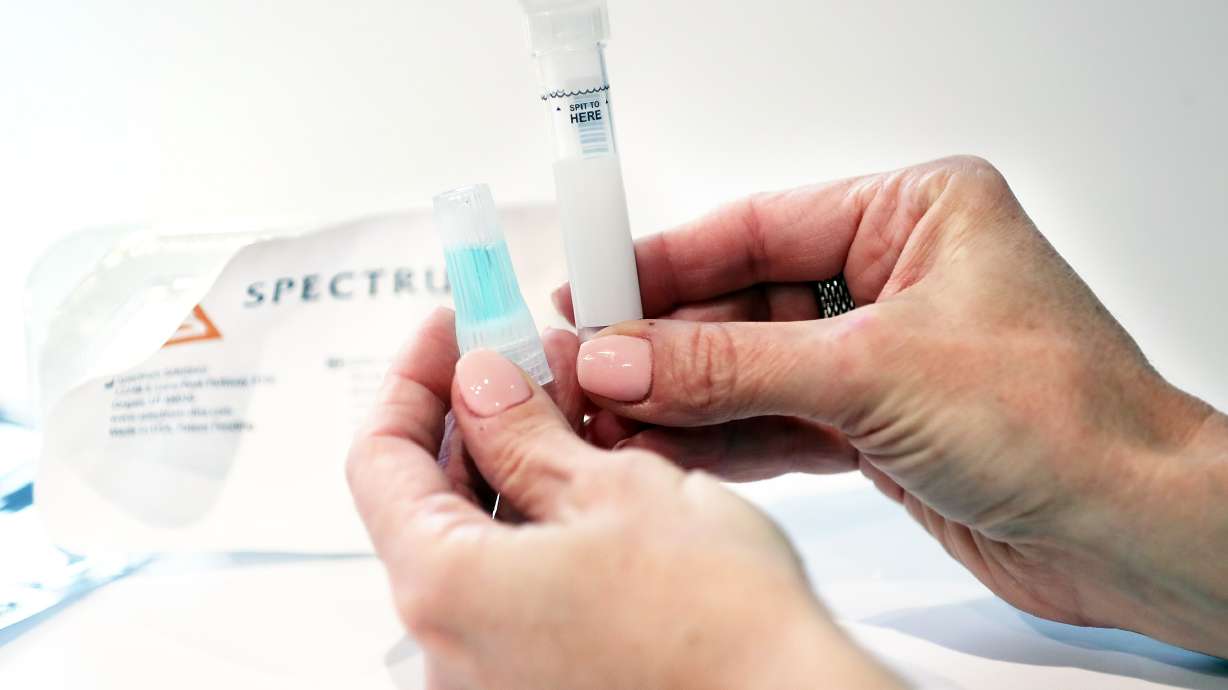
Researchers at Utah’s ARUP Laboratories and University of Utah Health have found that collecting saliva samples result in the same, if not more detection of the disease, said Dr. Kimberly Hanson, an infectious disease doctor at the U. and section chief of clinical microbiology at ARUP.
“It’s easier to collect and seems to be safer for the provider,” she said, adding that having a patient spit saliva into a tube requires less personal protective equipment and is less likely to produce the aerosols that are believed to transmit the novel coronavirus.
The study, representing one of the largest prospective specimen type comparisons to date, is published in the Journal of Clinical Microbiology. Other studies, including from the Yale School of Public Health, have reached similar conclusions but with markedly fewer patients and specimens.
For their study, Hanson said they recruited 368 volunteer patients who visited the Redwood Clinic’s in-car COVID-19 testing site for a COVID-19 test from late May through June. In addition to the nasopharyngeal swab test, which is successful at detecting respiratory illnesses (including COVID-19), the patients were asked to spit into a small plastic tube, providing enough to be tested at ARUP laboratories. They also tested another method using swabs at the front of the patient’s nostrils as well, which wasn’t as effective.
A couple dozen patients invited to participate could not, for whatever reason, produce sufficient saliva, Hanson said, but for most people, it was a completely viable test.
The tube was then placed in a plastic bag and handed back to the health care worker in “a simple and fast procedure,” Dr. Julio Delgado, chief medical officer at ARUP, said. He said the health care worker could essentially stand 6 feet away from the patient or behind a plexiglass window to collect the sample.
SARS-CoV-2 was detected in at least two specimen types for 90% of the patients who tested positive with the virus.
Of the 350 tested with both tests, the nasopharyngeal test detected 80 positive infections, while saliva detected 81. Neither test detected all infections, Hanson said. The nostril swabs missed nearly 15% of infections, according to the study.
“We know that no test is perfect,” Hanson said, adding that the virus was not always detected by both tests.
ARUP runs between 1,500 and 1,800 of COVID-19 specimens collected each day in Utah, varying from day to day, said Dr. Richard Orlandi, chief medical officer for ambulatory health at University of Utah Health. It is about a quarter of the total number of tests submitted to Utah laboratories on a daily basis in the state.
“It’s going to be a real game changer for how we’re testing suspected COVID-19 patients in Utah,” said Kathy Wilets, director of public relations at University of Utah Health.
Hanson said patients can expect results from a saliva test within 24 to 48 hours, which is similar to the nasopharyngeal swab that is commonly used for COVID-19 testing throughout Utah. But, she said, for severely compromised patients, results can be achieved as soon as eight to 10 hours.
Orlandi said he expects the saliva testing option to be available to patients no later than mid-September, but hopefully sooner.
“From the start of the COVID-19 pandemic, ARUP has worked to build capacity for high-quality COVID-19 testing,” Delgado said. “Our goal is to make this testing available to hospitals and health care systems nationwide.”